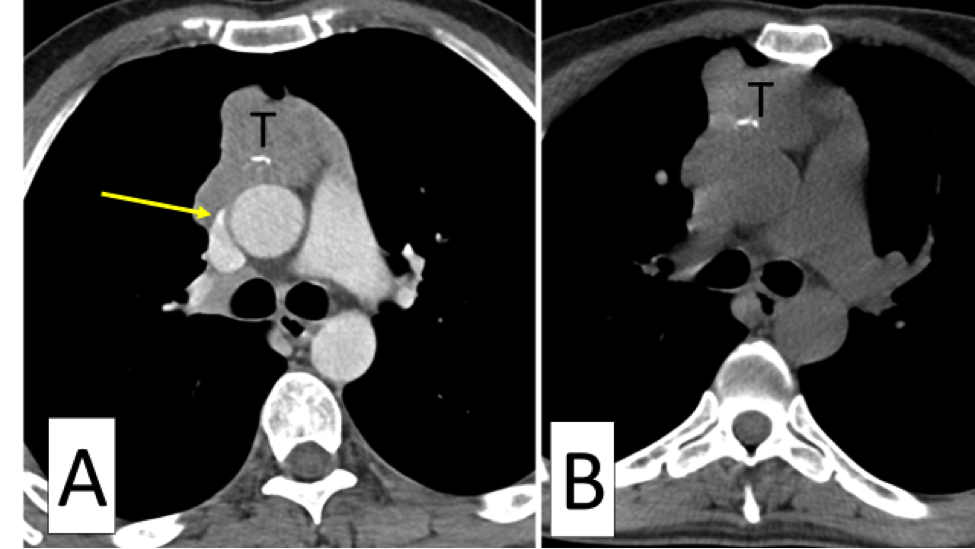
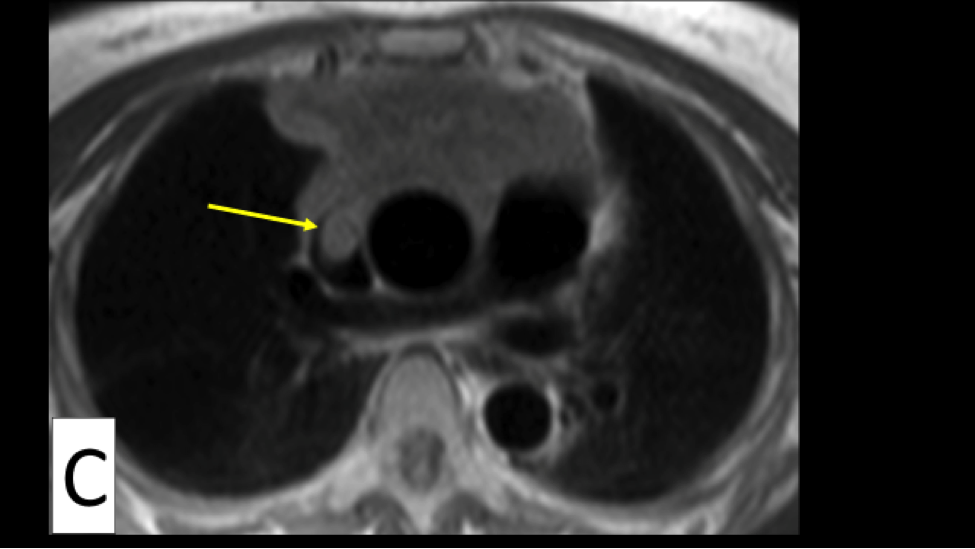
What’s your view if only immunotherapy (no surgery) has been used for stage 4A with lung pleural effusion and nodules? After 40 infusions, lungs appear clear with CT scan.
AR: Use of immunotherapy for previously untreated thymic cancers (i.e., no prior chemotherapy) would not be considered standard therapy.
If immunotherapy has been used for a period of two years or more in this setting and has been associated with a very good partial response or a complete response, it would be reasonable to stop treatment and continue with active radiological surveillance with CT scans approximately every three months. Treatment can be resumed if there is evidence of disease activity on follow-up scans. This paradigm of stopping immunotherapy after two years in case of a very good response and restarting it if there is evidence of disease activity, is increasingly used for treatment of lung cancer.
In regards to pregnancy and thymoma, Dr. Rajan spoke of non active disease and it being safe; however, I wondered about having active disease, not currently in treatment and whether it is ok to get pregnant although with the uncertainty of future scans? I have had some recent growth in a nodule within the past month as I was working with a fertility specialist.
AR: First, chemotherapy and other systemic anti-cancer therapies should be avoided during pregnancy, especially during the first 12-14 weeks, which is a critical period for fetal development. In situations where initiation of anti-cancer therapy is absolutely unavoidable, treatment is delayed until after the first trimester.
In this particular situation, if thymoma has been generally stable for months or years, the chances of significant growth within a period of a few months is low (especially, if histology is WHO subtype A, AB, B1 or B2). However, this is not guaranteed since the rate of growth can change over time. Therefore, although it is theoretically possible to have an uneventful pregnancy in the presence of quiescent disease, there is a risk that the rate of growth might increase during the course of pregnancy and additional treatment might become necessary.
If there is only one nodule growing within the past month and all other sites have a history of stability over a long period, one option is to consider local therapy for the growing nodule to treat it adequately and achieve overall disease stability before pregnancy.
For treatment of a solitary site of site of growth during pregnancy, surgery would carry with it risks of exposure to anesthetic medications. Radiation therapy should be avoided. Another option is thermal ablation (cryoablation or microwave ablation), which might be relatively safe under these circumstances and has been used to successfully treat solitary sites of active thymoma.
Ultimately, this is a complex question and requires a detailed discussion between a patient, her obstetrician and the oncology team.